Matrix Natural History Study: Snapshots at Year 1

Hello everyone and welcome to Science with Sandra!
This time, I would like to share with you the preliminary results of the MATRIX Natural History Study that launched in July 2022.
This report is just preliminary and we cannot make any solid conclusions yet, however I wanted to share this brief snapshot so that you can see the type of questions that have been asked and the information the participants have shared anonymously. I hope this report brings awareness of the importance of the participation of our global community and the benefits it will bring to our community as a whole.
The goal of this study is to help understand the individual experiences of people who have this disease so researchers can better understand its impact on patients and how that may change over time. These insights will be critical for developing new and better ways to diagnose and treat Glut1 Deficiency.
The method for this study is the use of patient reported surveys. The virtual surveys being implemented are developed by ClinGen to capture a snapshot of a person’s overall health and be able to monitor for changes over time.
The short surveys use common data elements and can be compared across other diseases. They are available in English, Spanish, Italian, French, and German with Brazilian Portuguese, Hebrew, and Korean coming soon.
Participants start with a general health and development “head to toe” survey of all body systems, and any issues noted in specific areas then branch into more detailed, in-depth surveys to capture more information.
Surveys will be repeated each year to capture changes over time.
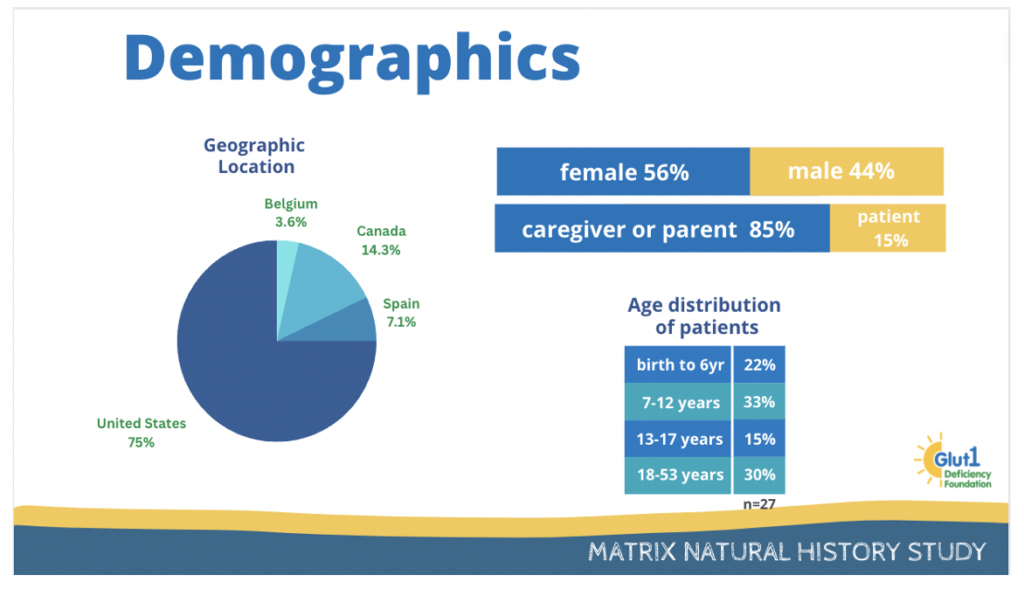
The results we have obtained until now are shown below. Regarding demographics, 30 patients from around the world have registered to participate in this study, however, some of them have not completed the consent form to participate in the study, some have not uploaded their genetic test or lumbar puncture results to confirm the diagnosis and some have not completed the surveys.
In regards to the genetic information, 16 patients have uploaded their genetic tests and the summary of the information reported is below.

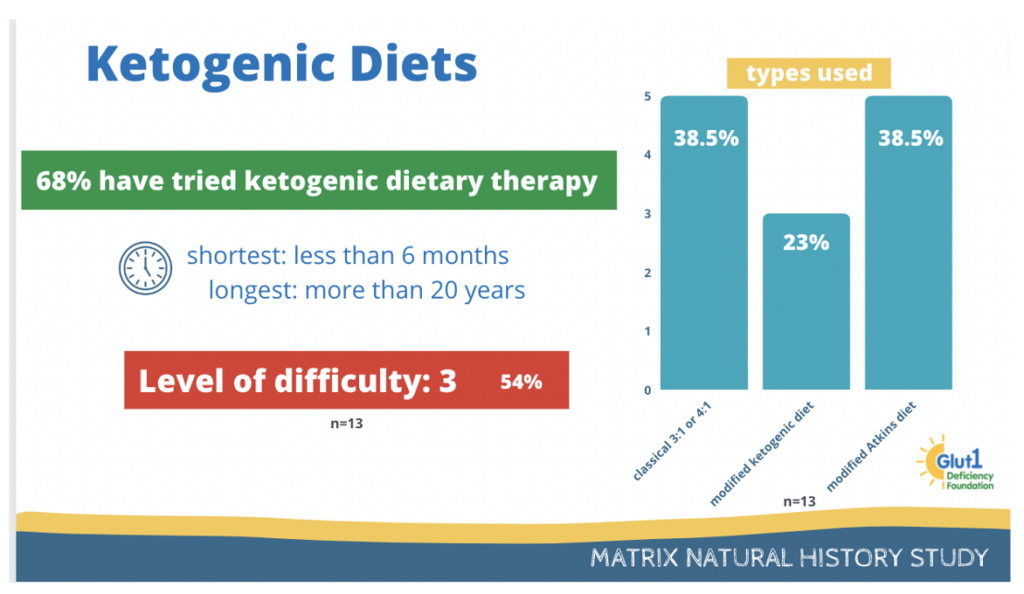
Participants responded a brief survey about the ketogenic diet and you can see the results below.
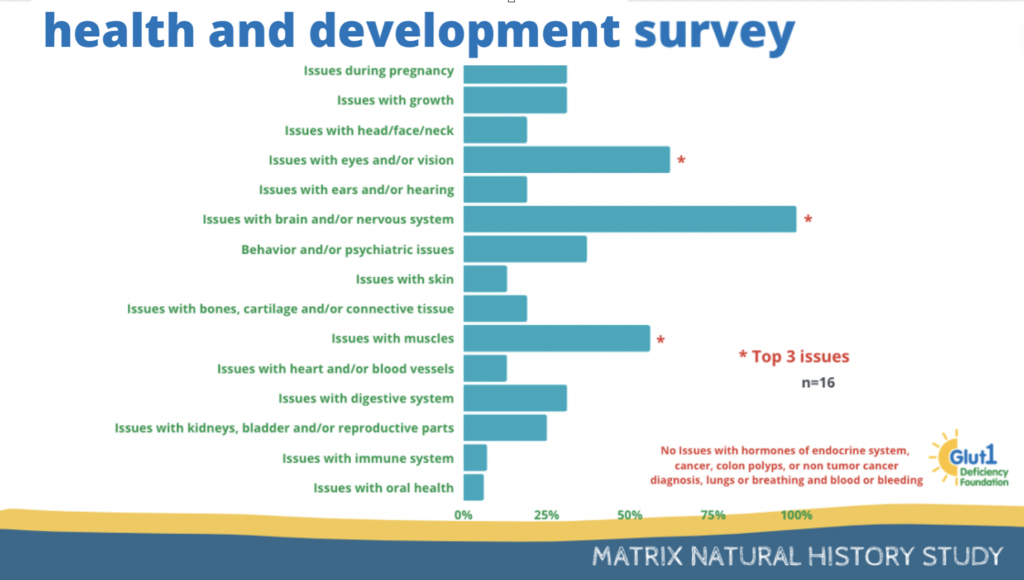
Results of a survey about general health and development can be seen below.
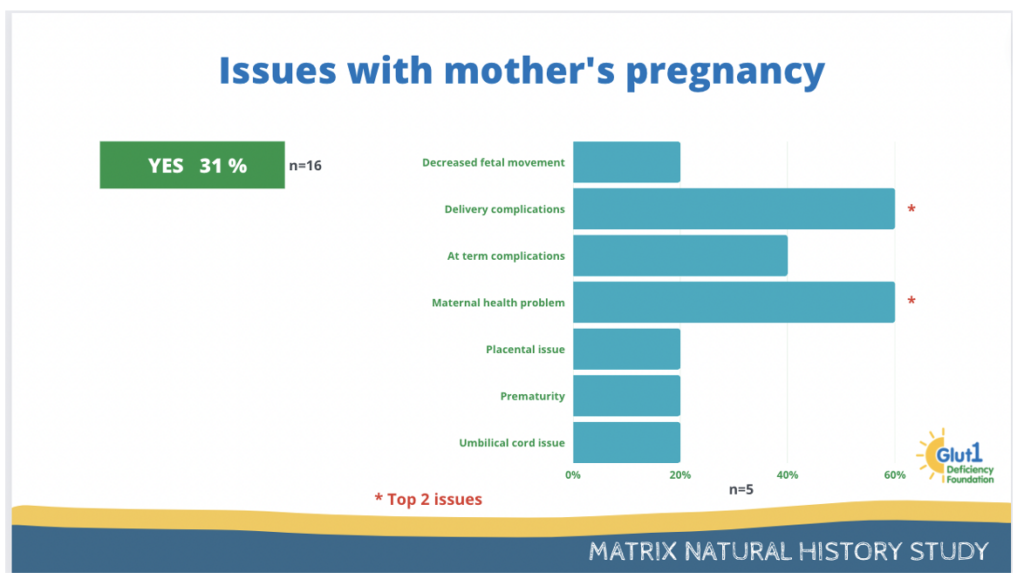
31% of the participants reported having issues with mother’s pregnancy.
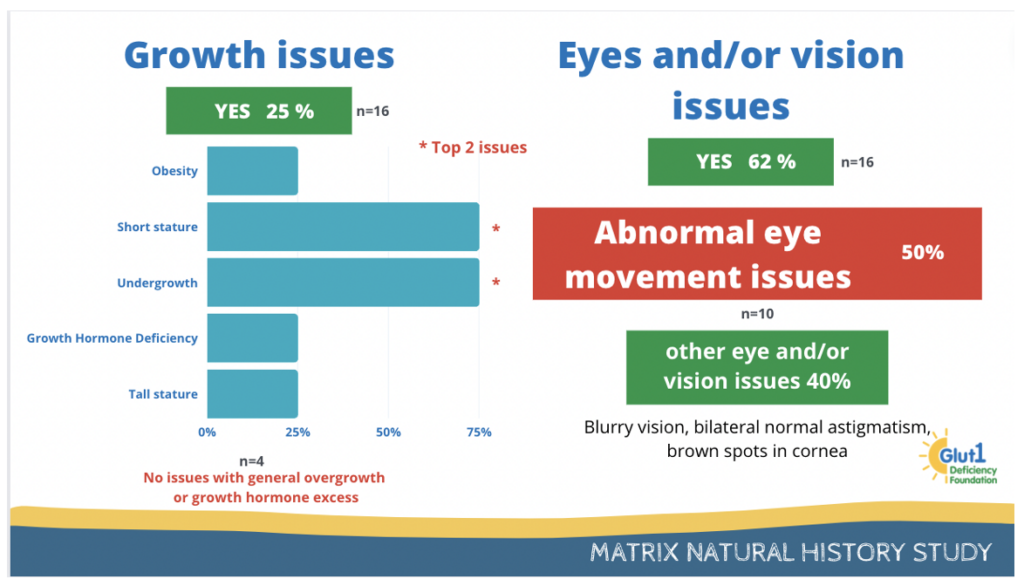
25% of the participants have reported issues with growth and 62% have reported issues with eyes and or vision.
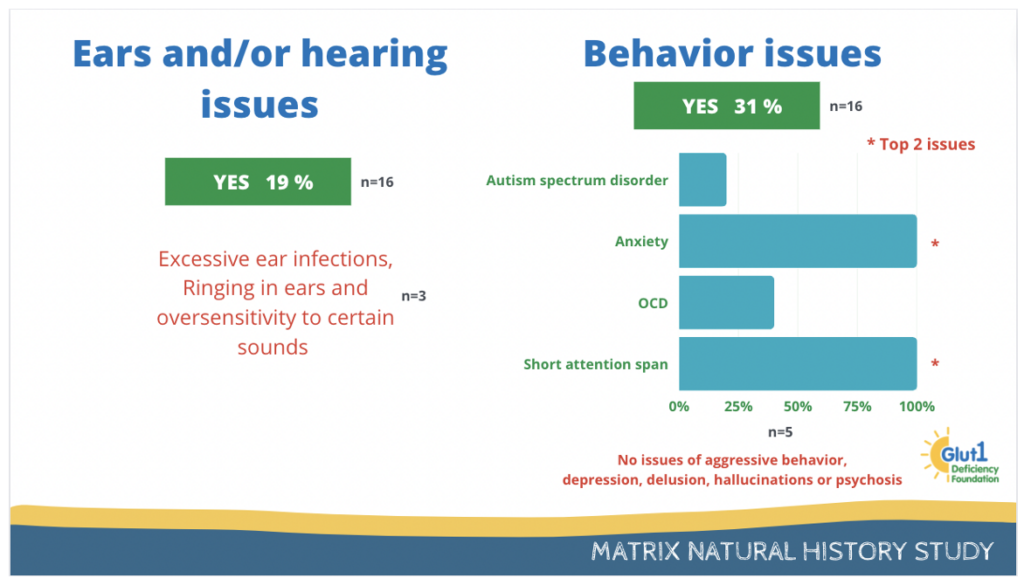
19% have reported ears or hearing issues and 31% have reported behavior issues.
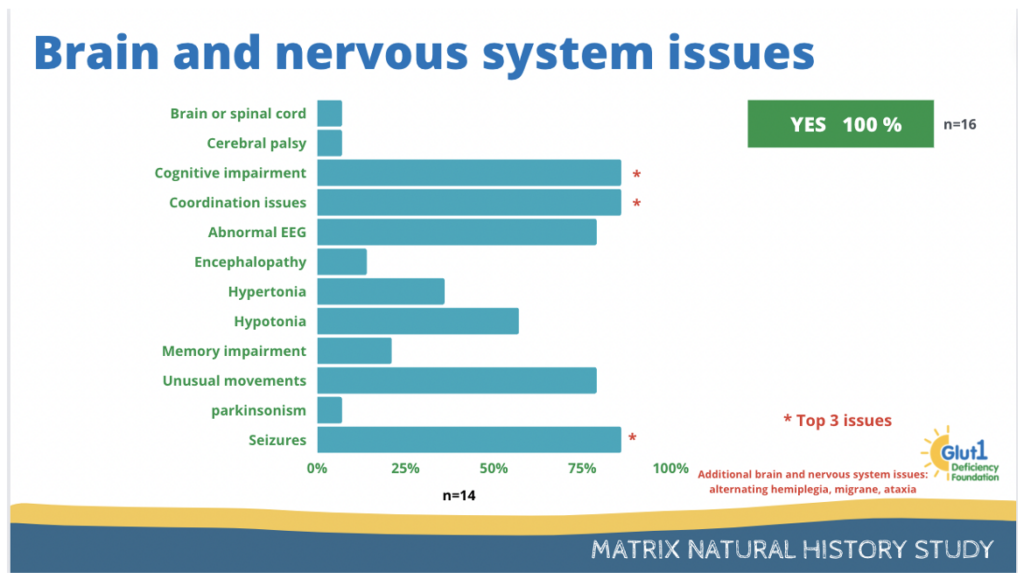
All participants have reported issues with brain and the nervous system.
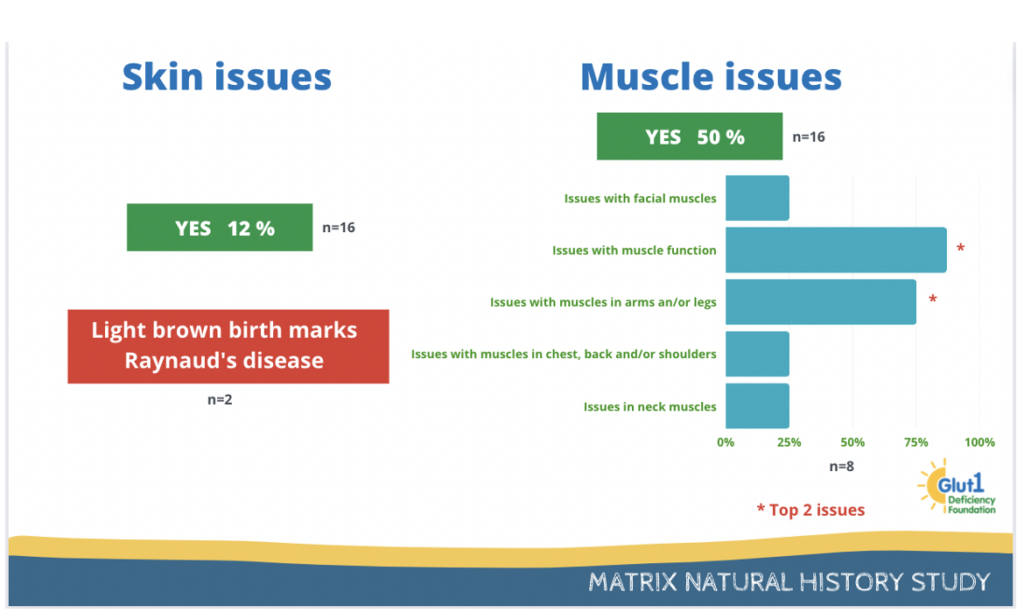
12% of participants report having issues with skin and 50% report having muscle issues.
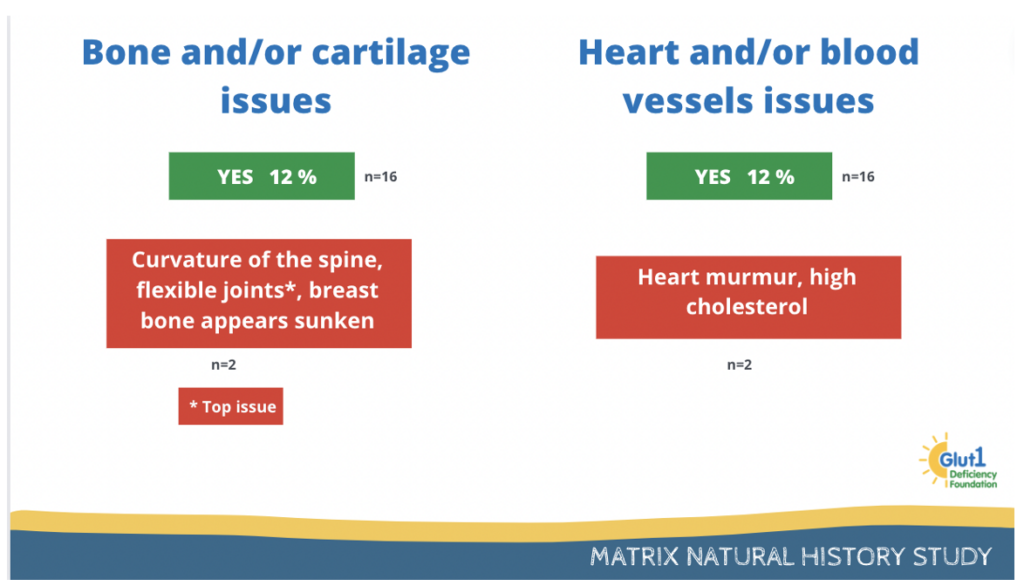
Bone and cartilage issues as well as heart and/or blood vessels issues were reported by 12% of participants.
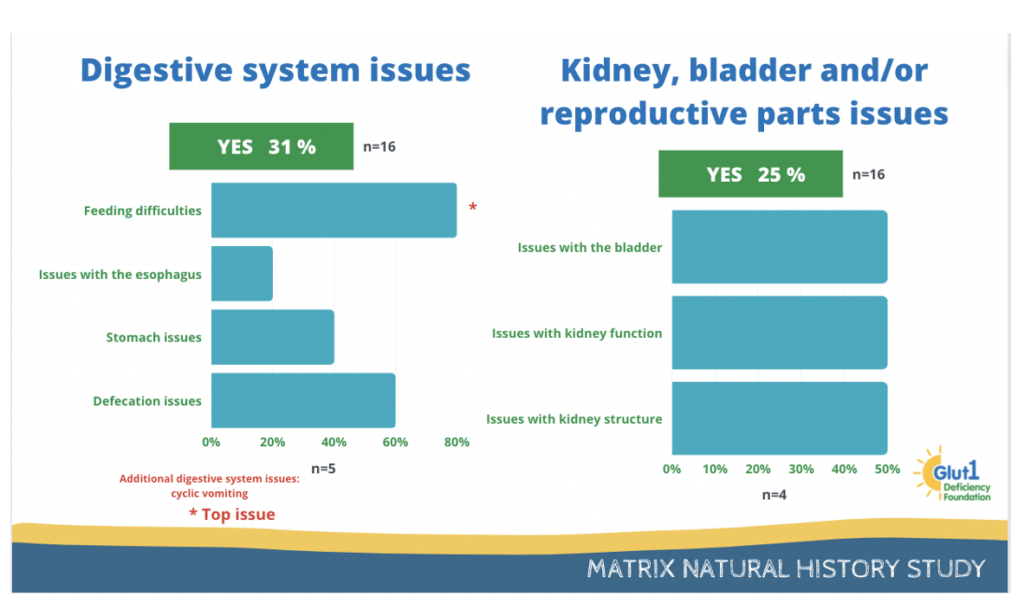
31% of participants report having issues with the digestive system, and 25% report having issues with kidney, bladder and/or reproductive parts.
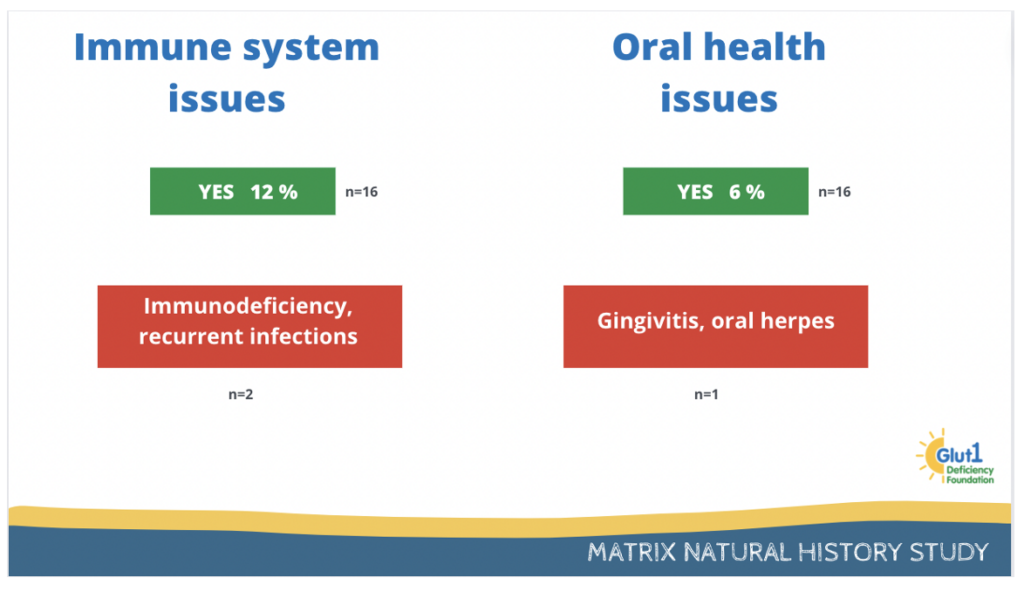
Finally, 12% of participants report having immune system issues and 6% report having oral health issues.
The discussion:
- The data presented here is just a glimpse of a small group of patients’ reported data
- The number of participants is too small to make any conclusions
- More patient participation is needed in order to have more robust data that could be used to develop better treatments and to be used potentially as the placebo arm in future clinical trials
If you would like to participate in this study you can find more information in this link, or you can email me to [email protected].
Please do not hesitate to contact me if you have any questions or comments, or if I can be helpful to you in setting up an account and completing the surveys. We encourage all families to participate and ensure their loved one with Glut1 is included in these disease snapshots.
We are very grateful to those who have participated already and have told your individual story of Glut1 Deficiency through the data you contributed.
Thank you!



