About Glut1 Deficiency
Glucose Transporter Type 1 Deficiency Syndrome
also known as G1D, Glut1DS, De Vivo Disease
Glut1 Deficiency is a rare genetic condition that impairs brain metabolism. It is caused by mutations or variants in the SLC2A1 gene, which encodes the glucose transporter protein type 1 (Glut1). Glut1 is the principal transporter of glucose and also moves other important sugars across the blood-brain barrier. Glucose is the primary source of energy for cells and is critical for many metabolic processes.
Impaired glucose transport associated with Glut1 Deficiency creates a metabolic crisis in the brain and often results in a range of neurological symptoms such as seizures, speech and movement disorders, and developmental delays. Not all patients experience all symptoms, and there is a wide spectrum of severity. Symptoms may change and evolve over time.
The standard of care treatment is a ketogenic diet. This low carbohydrate, moderate protein, high fat diet causes the body to produce ketones, which can be used as fuel by the brain and other tissues when glucose is limited. The diet helps most patients with most symptoms, even in adulthood, but it is not a cure. It is thought that ketogenic diets help preserve brain growth and development in Glut1 Deficiency, so early diagnosis and treatment is critical.

Glut1 Deficiency
Symptoms
The hallmark symptoms of classical Glut1 Deficiency are seizures (approximately 90% of patients), a complex movement disorder, developmental delay and intellectual disability, and speech/language impairments. Not all patients experience all symptoms, and there is a great deal of variance across a wide spectrum in both the combination and severity of symptoms from one patient to another. Symptoms and treatment response can change over time.
Retrospective natural history studies have shown that, in general, seizures improve in adulthood and movement disorders become more pronounced. Puberty often brings changes in symptoms.
Suggestive symptoms seen in many Glut1 Deficiency patients:
early onset absence seizures
typically starting in the first year of life but sometimes in later childhood; however, patients can have many different types of seizures and 10 to 15% do not have any
unusual eye-head movements
typically starting in infancy and resolving in early childhood; parents report this is often the first symptom indicating something is wrong
treatment resistant seizures/epilepsy
at any age
complex movement disorders
often unique forms and combinations of ataxia, dystonia, chorea, gait abnormalities, etc.
fluctuation of symptoms
hunger, fatigue, heat, illness, anxiety, excitement and other triggers; symptoms often worse first thing in the morning and can improve after a meal
episodic neurological symptoms
seizures, movement disturbances, hemiplegia, headaches, energy levels, confusion, mood
PED episodes
Paroxysmal Exercise Induced Dystonia/Dyskinesia
developmental delays
global impacts on all domains of development
intellectual disabilities
broad range of cognitive impacts
complex speech and language disorders
articulation, fluency, oral motor coordination, processing
Other conditions sometimes diagnosed in patients with Glut1 Deficiency:
more insights
Collective Voices Project
Learn more about symptoms and patient and family experiences, priorities, and needs from our patient voice survey done in 2021.

watch our latest
Educational Videos
Diagnosing Glut1 Deficiency
Lumbar Puncture:
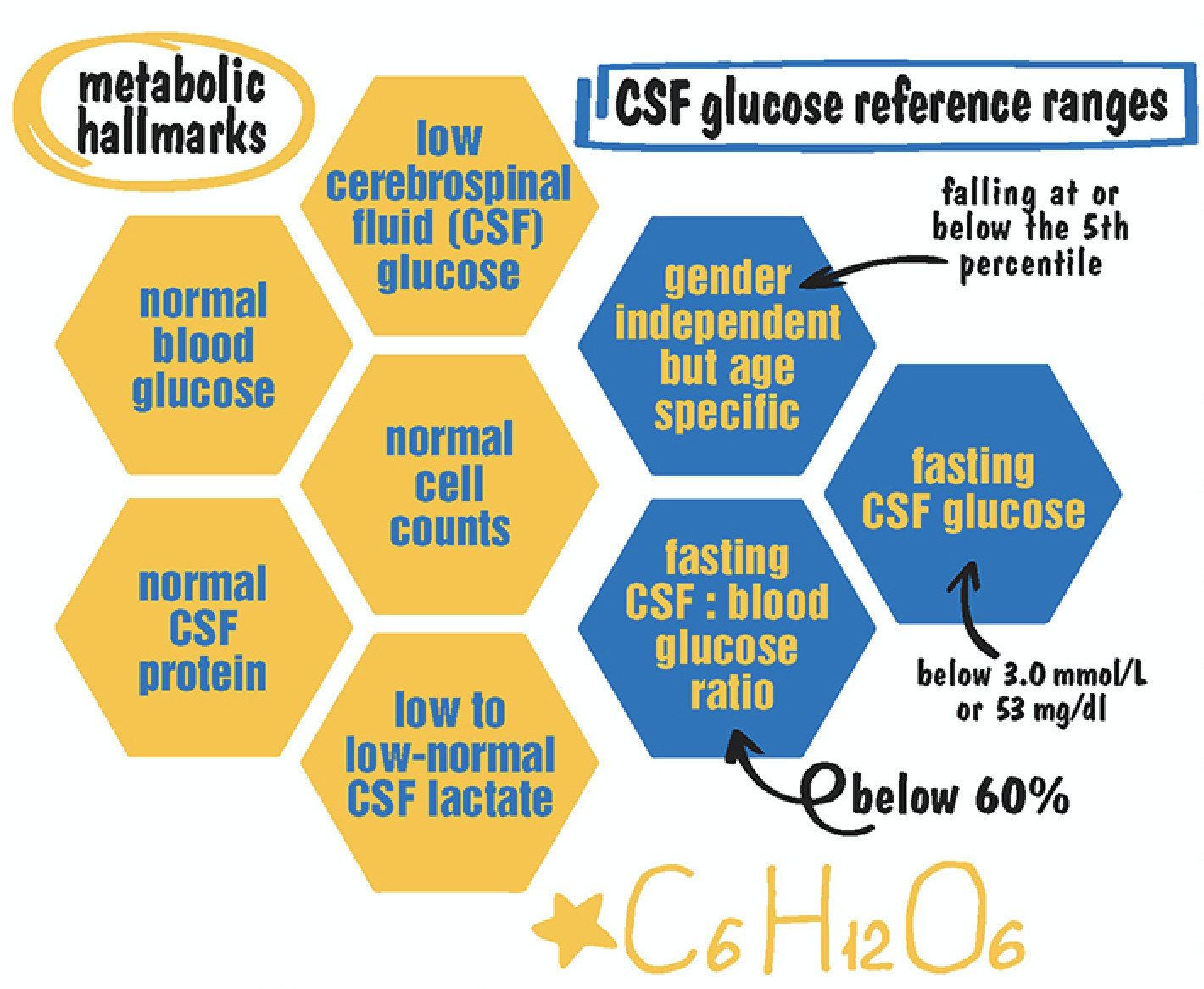
When Glut1 Deficiency is suspected, glucose should be measured in the spinal fluid and in the blood simultaneously after a 4-6 hour fast. Blood samples should be drawn first to avoid stress-related hyperglycemia, and a lumbar puncture (spinal tap) should quickly follow.
Metabolic Hallmarks of Glut1 Deficiency:
Glucose Reference Ranges in Glut1 Deficiency:
Genetic Analysis:
Genetic testing can also help confirm the diagnosis by detecting a mutated SLC2A1 gene, although current testing doesn’t identify a mutation in 10-15% of patients. The combination of suggestive clinical symptoms and the characteristic CSF findings indicate a Glut1 Deficiency diagnosis, even in the absence of an identified SLC2A1 mutation.
Distinct patterns of brain glucose uptake on PET scans and specialized red blood cell uptake assays are also useful in the absence of an otherwise clear diagnosis but might be available in specialized centers only.
Dystonia 9 and 18 are associated with mutations in the SLC2A1 gene.
Other Tools:
Metafora Biosystems is a company based in France that is developing a blood test, METAGlut1, to diagnose Glut1 Deficiency through detecting cell energy abnormalities. The test is currently available in France, Belgium, and Luxembourg with more research and development underway to make it more widely available.
Genetic Testing Resources:
Glut1 Deficiency
Treatment
There is currently no cure for Glut1 Deficiency, but the recommended standard of care treatment is a medically supervised ketogenic diet, which can improve most symptoms for most patients, even in adulthood. A ketogenic diet is a high fat, moderate protein, low carbohydrate diet that provides alternative fuel to the brain with ketones instead of glucose. The earlier the diet is implemented, the better the outcomes for patients in improving seizure control, movement disorders, and cognitive abilities. Studies show that 4 out of 5 children who have epilepsy caused by Glut1 Deficiency will become seizure-free on dietary therapy.
In infancy and childhood, a classical 3:1 or 4:1 ketogenic is recommended to ensure the highest level of ketones to the meet the metabolic fuel demands of the developing brain. Although it is recommended to continue the classical ketogenic diet for as long as is tolerated by the patient, alternative ketogenic diet versions such as the Modified Ketogenic (2:1 and 1:1 ratios) or the Modified Atkins Diet may be more feasible for quality of life and compliance considerations and are often used by teenagers and adults.
All patients on a ketogenic diet should be under the care of an experienced dietitian and neurologist and have regular laboratory screenings to help monitor for potential side effects. Blood ketone levels, as opposed to urine, should also be monitored and can be correlated to optimal brain energy supply and symptom control.
For a small subset of patients, a ketogenic diet proves ineffective despite adequate levels of ketosis, which illustrates that there is much still to learn and new treatment options are critical and urgent.
Medications to address the symptoms of seizures or movement disorders may provide benefit, although there is currently no clear basis for specific recommendations and there are concerns to consider regarding potential harmful interactions with ketogenic diets.
Prevalence
The number of patients diagnosed is currently thought to number approximately 2,000 worldwide. Recent studies have estimated true prevalence to be at least 1:24,000, so the vast majority of patients remain undiagnosed. There’s no known susceptibility related to gender or race.
Frequently Asked Questions
Glut1 Deficiency is caused by variants or mutations in the SLC2A1 gene, although not all patients will have a genetic variant detected on current testing. The SLC2A1 gene is responsible for producing and regulating the glucose transporter type 1 protein (Glut1). Glut1 is responsible for moving glucose across the blood brain barrier. Glucose is a primary source of energy for all cells and plays a vital role in brain metabolism. When there isn’t enough Glut1 or it isn’t functioning properly, glucose doesn’t reach the brain as well as it should, and many neurological symptoms result.
There is a wide range of symptom severity, and not every person with Glut1 Deficiency experiences the same symptoms, so this makes it difficult to predict what an individual experience and future might hold. Some patients are very mildly affected with just a symptom or two and are able to lead an independent life, while others are quite severely impacted and might be unable to walk, talk, or function independently. Most fall somewhere in between.
It is also important to remember that less is known about the adult experiences in Glut1 Deficiency, especially later in life. Symptoms and severity do not seem to be progressive over the lifespan, although there can be fluctuation in severity and change in symptoms as patients age. There is no known impact on life expectancy, although there are increased risks associated with any type of epilepsy for seizure related deaths or SUDEP (Sudden Unexplained Death in Epilepsy).
The good news is that most people with Glut1 Deficiency, regardless of age, respond well to a ketogenic diet and experience improvement in many symptoms. There are many ketogenic diet resources and links available on our website.
There is no cure for Glut1 Deficiency, so treatment is aimed at managing symptoms. The current standard of care is a medically supervised ketogenic diet. Ketogenic diets are high fat, moderate protein, and low carbohydrate diets that cause the body to produce ketones, which the brain can use as a fuel source when glucose is limited. There are likely other mechanisms at work in explaining how ketogenic diets can help the symptoms of Glut1 Deficiency, but those are not yet well understood.
Research has shown that the earlier someone is diagnosed with Glut1 Deficiency and begins treatment with a ketogenic diet, the better the outcomes. There is research underway to identify other options for patients and families for easier and more complete treatments as well as for those who many not respond well to a ketogenic diet.
Additionally, other types of support are beneficial in treating Glut1 Deficiency, such as speech and language, occupational, and physical therapies. Special supports and plans in place in the classroom (such as IEP’s, Individualized Education Plans, and 504 plans) are also very helpful in optimizing academic success and the overall school experience, and special accommodations in the workplace can also be important forms of support.
Glut1 Deficiency is a rare disease, and currently we believe there are about 2,000 people diagnosed worldwide. Without a formal way to report and track patients, it is very difficult to know exact numbers, and that is one of the reasons why projects like the Glut1 Deficiency Natural History Study is so important. Recent research has also estimated that the true prevalence is at least 1 in 24,000, which means there are many more people still to find who need a life-changing diagnosis.
Take some time to come to terms with this new diagnosis and what it means for you and your family. It often comes as a big relief to have answers, but there can also be a grieving process that is important to acknowledge. Once you are ready, there are many ways you can empower yourself on this new journey and put your hope for a brighter future into action. Look through our many resources to find information and opportunities to help you better understand this disease and help you be an advocate for you, your loved one, and everyone in the Glut1 Deficiency community.
Most cases of Glut1 Deficiency are caused by a de novo variant or mutation, which just means it is a new, spontaneous one that happened for the first time with no known cause. This means that neither parent has the genetic variant and it wasn’t inherited. However, when someone is affected by Glut1 Deficiency and has a genetic variant, it can be passed on to offspring with each child having a 50% chance of inheriting the disease from their parent. It is an autosomal dominant inheritance pattern, which means it only takes one mutated copy of the gene (out of two) to cause disease.
Talk to your doctors about your diagnosis and create partnerships when possible. You will be the expert in many circumstances, so it is important that you understand this disease well, keep up with new advancements and research, and feel confident in being an advocate for yourself or your child. The Glut1 Deficiency Foundation can send brochures for you to share with your healthcare team, and the expert consensus guidelines are another helpful resource to share. You can also send them to this website for additional resources, and even invite them to join our Professional Network and attend one of our conferences!
We have created a directory of healthcare providers who have been recommended by Glut1 Deficiency families for their experience, compassion, and the quality of care they provide. You may find that Family Recommended Specialist List here. It is also important to remember that a willingness to learn about a disease and a commitment to caring for patients well can be a good substitute for experience, so you can help create the next generation of Glut1 Deficiency experts through partnerships and collaborations with your healthcare team.
The Glut1 Deficiency community is like a family, and you’ll find many who are eager to welcome you and help you on this new journey. We have created a secure, private forum where you can connect with other families, ask questions, share ideas, and give and get support – Glut1 Community Connect on the Discourse platform. You’ll receive an invitation to join when you sign up for the Family Network.
You can also join our monthly Glut1 Gatherings on Zoom to learn and meet families from all over the world. Teens and adults with Glut1 Deficiency have their own groups that meet monthly on Zoom, too. Every other year, we host our conference where we meet, share, and learn with patients, families, clinicians, researchers, and other stakeholders who are working hard to help us all reach for a brighter future.
PATIENTS, FAMILIES & CAREGIVERS
Explore how Glut1 Deficiency can affect each stage.
Infants and Toddlers
Typically, most Glut1 Deficiency symptoms start to emerge in the first year of life, but that isn’t always the case. Unusual eye/head movements (aberrant gaze saccades or intermittent involuntary gaze) were reported by families as the most frequent first sign that something was wrong. These resemble opsoclonus or nystagmus and are sometimes mistaken for them, but the head movement component makes them unique and thought to be distinct to Glut1 Deficiency. They often appear in infancy and typically stop by 5 or 6 years of age.
Seizures are also a frequent first symptom, most often appearing in infancy. These can be any type of seizure, but absence and myoclonic tend to be the most common and can sometimes be hard to recognize. There are other symptoms that may be difficult to recognize, too, until later, such as developmental delays or intellectual difficulties. Issues with muscle tone and balance are common in this stage and sometimes cause delays in reaching milestones like rolling over, crawling, walking, and toileting. Speech and language development may not be typical.
Children diagnosed at this age with Glut1 Deficiency can expect to be put on the classical ketogenic diet (a 3:1 or 4:1 ratio), which helps ensure optimal symptom control for most and helps preserve brain growth and development. Ketogenic formulas are readily available commercially, and it is possible to integrate breastfeeding into a ketogenic diet plan. Early intervention programs can help provide support for different therapies and services that may be beneficial.
Children
Children with Glut1 Deficiency can experience new symptoms at this stage, including new seizure types and new movement episodes. Ataxia may become more noticeable and so can spasticity and other involuntary movements. Some begin to have temporary hemiplegic episodes that can mimic a stroke, which can be particularly frightening but important to recognize and understand as part of the symptom picture for some patients.
Developmental delays and cognitive disabilities can become more recognizable at this stage, especially as children with Glut1 Deficiency enter school and their challenges become more apparent. Speech and language disorders may continue to emerge, and attention and focus issues can negatively impact school performance. Early recognition of these challenges helps get appropriate support and accommodations in place, especially in the school and classroom setting, so that children with Glut1 Deficiency can find academic success, social acceptance, and lay good foundations for future school experiences.
Children diagnosed at this age with Glut1 Deficiency can expect to be placed on a classical ketogenic diet in most cases, but sometimes also a Modified Ketogenic Diet with a lower ratio. Early childhood is a period of rapid brain growth and development, the most intense in any stage of life, and ketogenic dietary therapies can help improve outcomes. Physical, occupational, speech/language, and other therapies can be beneficial in either the school or private practice setting.
Adolescents & Teenagers
Adolescence can be a time of change and a time of challenge for Glut1 Deficiency patients. New symptoms can emerge, including new seizure types, and sometimes movement disorder episodes begin to happen for the first time or new types are experienced. Families may notice that there are triggers for these episodes, such as prolonged physical exertion, hunger, fatigue, illness, warm temperatures, and others. Anxiety can become more pronounced and social challenges can emerge around differences and inclusion, both from the impacts of the disease as well as the realities of following a ketogenic diet.
Ketogenic diets are still the recommended treatment for this age, but some experience reduced effectiveness or challenges in maintaining ketone levels as high as they need to be to experience the same benefits as in the past. Compliance can also begin to become more of an issue, so sometimes less restrictive versions of the diet, such as a Modified Atkins Diet may be tried that allow more flexibility. Some adolescents and teens may still need a Classical Ketogenic Diet to have the best symptom control. Various therapies can continue to be beneficial, and opportunities to participate in group programs and have extra social support can be helpful.
Adults
Although much remains to be understood about the adult experience in Glut1 Deficiency, especially mid-life and senior years, it can be a continued time of change and challenge. For some adults, seizures can improve or even disappear, but for others, new seizure types can begin for the first time. Movement disorder episodes can increase in both severity and frequency, and new types can begin to occur as well. These movement episodes are especially challenging as they don’t always respond well to current treatments. Energy and stamina issues are also frequently reported.
Opportunities for social engagement and participation in regular routines become more difficult in adulthood once the formal school experience comes to an end, so it is important to plan early for this stage in life to help bridge the supports and opportunities found at school with new ones to take their place. Although ability levels do vary widely, this is a stage when the lack of independence caused by this disease can have the most impact on quality of life, and it is important to consider and plan for the long-term support that people with Glut1 Deficiency might need across their lifespan. Research suggests that those who have been on a ketogenic diet do not experience loss of skills over time as they age, but there is little knowledge and experience in later adulthood. Life expectancy is not known to be impacted directly, but there are increased risks associated with any form of epilepsy for seizure-related deaths or SUDEP (Sudden Unexplained Death in Epilepsy).
Ketogenic diets can still be very beneficial in adulthood, even for those who do not receive their diagnosis until this stage of life. Compliance and lifestyle challenges can sometimes make a Modified Atkins Diet or Modified Ketogenic Diet more feasible at this stage. Physical, occupational, and speech therapy are also still beneficial, and many families report that moderate exercise can also help with symptoms. Transition of care from pediatric to adult medical providers is also an important process to plan for in advance.

